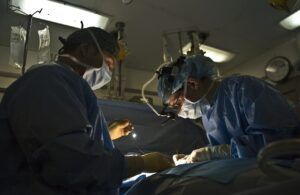
Defective Medical Devices: Malpractice or Product Liability?
When defective medical devices injure people, there is often a key question that needs answering: is it a matter of medical malpractice, or do you have a product liability claim on your hands?
Defective medical devices pose a threat to patients’ health. When the medical tools and equipment don’t work as intended or are improperly used, it can cause harm to a patient. When medical devices injure people, there is often a key question that needs answering: is it a matter of medical malpractice, or do you have a product liability claim on your hands?
What’s Considered a Medical Device?
A medical device refers to any tool, machine, equipment, instrument, or implant used throughout the course of diagnosis and/or treatment. This definition encompasses a variety of medical devices, from a stethoscope to complex prosthetics. When medical devices don’t work as intended, it can cause injuries, like scarring or infections, or death. Some examples of types of medical devices that commonly cause issues are stents, implants, defibrillators/pacemakers, and contraceptive devices.
Medical Malpractice Claims
In some cases, negligent care from healthcare providers causes medical devices to harm a patient. The hospital or specific staff member can be held responsible for providing negligent care. A doctor could have failed to inform the patient on how to use the medical device or failed to monitor the device’s function after administering or implanting, which led to worsened health conditions. Other medical malpractice claims related to defective medical devices may include implanted devices that cause an infection or are improperly placed and rejected by the body. Doctors can also be held responsible for knowingly using a defective device. A medical malpractice attorney could investigate the circumstances surrounding your case.
Product Liability Claims
In other cases, it is not the health care provider’s negligence, but the product itself that causes injury. Such claims involving defective medical devices can be classified into three types of product liability claims:
- Manufacturing defects: Improperly manufactured devices that were either made incorrectly or sustained damage during manufacturing or transportation to the user.
- Design defects: When the product poses a foreseeable risk of danger, even when it’s being used it for its intended purpose. In other words, the product is unreasonably dangerous despite being manufactured properly . A non-medical example would be the recent example of certain smartphones with batteries that sometimes overheated and caught fire. The problem was the way the battery was designed, not an error in manufacturing the battery. The producer did not forget to attach a certain piece to the phone. The thickness of the barrier around the flammable battery was insufficient. Thus, liability in that case would be based on showing that the entire line of smartphones, as designed, created an unreasonable risk to the consumer.
- Marketing defects: Misleading, false, or omitted information, such as warnings and instructions concerning a medical device. This causes the health care provider or patient to use the device incorrectly or in a dangerous manner.
Contact The Law Offices of Cardaro & Peek, LLC Today
Do you believe that you or your loved one has suffered as a result of medical malpractice or medical neglect? You need to talk to an experienced team of local lawyers today.
The lawyers at Cardaro & Peek, LLC have the experience and resources necessary to investigate and litigate all types of medical claims throughout Maryland and Washington D.C. Cardaro & Peek, LLC has medical personnel on staff and has access to nationally recognized, board-certified physicians and other experts, to assist in the investigation, analysis, and prosecution of all types of medical malpractice claims. If you or a loved one have experienced malpractice, give us a call at 410-752-6166. Please visit our website www.cardarolaw.com and follow us on Facebook , Twitter , and LinkedIn for more information.
The post Defective Medical Devices: Malpractice or Product Liability? appeared first on .